Boxwood Dieback
Free printed copies of pest alerts are available.
Note: do not confuse boxwood dieback with boxwood blight (pdf download), which is currently much more common.
Introduction
Boxwood (Buxus sp.) is an important perennial landscape shrub in the United States. It is considered a top ornamental choice for new housing and other commercial development because of its vibrant green color and herbaceous evergreen growth.
According to the 2019 USDA National Agricultural Statistics Service, the wholesale market value of boxwoods was estimated at $130 million annually in the United States.
Boxwoods are known to be infected by a number of plant diseases that include boxwood blight, Macrophoma blight, Phytophthora root rot and Volutella blight. In 2015, a new disease of boxwood called boxwood dieback was identified in Louisiana and is the first known report in the United States (Singh et al. 2015).
Boxwood dieback is a foliar disease caused by a fungal pathogen, Colletotrichum theobromicola. Research suggests that an association probably exists between Colletotrichum spp. and boxwood (Crouch 2012; Farr and Rossman 2016; Holcomb 1967).
Symptoms
Boxwood dieback symptoms include random dieback of twigs with light tan colored foliage (Figure 1). Affected leaves do not defoliate and tend to stay attached to the branches.
Root and crowns of affected plants look normal (Figure 2). These symptoms on boxwoods have been long observed in landscape plantings but were always attributed to Phytophthora root rot or Volutella blight.
The infection also causes bright black discoloration of stem immediately under bark (Figure 3). This bright black discoloration extends all along the infected twigs and differs from discoloration of the crown region caused by phytophthora root rot.
Other biotic and abiotic agents that may cause similar symptoms to boxwood dieback include boxwood blight, Macrophoma blight, cold injury, and herbicide injury.
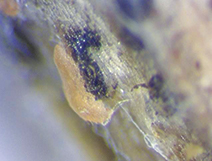
In addition to twig dieback, the pathogen also causes leaf spots (Figure 4) that appear white in the center surrounded by dark brown margins. Under favorable environmental conditions, black fungal fruiting bodies can be observed in the spots.




Geographic Distribution and Host Range
In the United States, boxwood dieback has been detected and identified in several states in the south and southeastern U.S. Complete information on the susceptibility of boxwood cultivars to C. theobromicola is not available. However, English, Japanese and Korean cultivars of boxwood have been found to be very susceptible.
Disease Epidemiology
Boxwood dieback is a foliar disease that has been detected from boxwood liners (Figure 5). The disease is thought to be introduced to new locations via infected liners.
The pathogen is known to produce spores on young, infected twigs in the landscape (Figures 6 and 7). Disease spread from plant to plant is accomplished by poor pruning practice, and by the dispersal of conidia via rain or irrigation water.
Although environmental conditions affecting disease development are not currently known, artificial inoculation of boxwood plants maintained in a greenhouse at 28 ± 2°C and 85% relative humidity has demonstrated that symptom development will occur after three months of incubation.


Disease Management
Since boxwood dieback is a recently discovered disease, effective diagnostic tools and control measures such as fungicides are currently limited. Landscapers, nurserymen and homeowners should follow good cultural practices and create an environment that will hopefully decrease the spread and development of boxwood dieback.
Since removing dead and dying twigs from plants infected by the pathogen is not known to control this disease, all symptomatic plants in the landscape should be removed and destroyed. Surface disinfection of pruning and cutting tools is important to reduce its spread. Avoiding unnecessary plant injury may also help avoid any potential infection by the pathogen.
Nursery owners should closely monitor liners and potted boxwoods for symptoms of boxwood dieback. Suspected plants must be immediately isolated from healthy plants.
Disease management strategies practiced for managing phytophthora root rot will not provide management of boxwood dieback. Laboratory testing is required to confirm boxwood dieback because it can easily be misdiagnosed as phytophthora root rot.
Sample Collection and Submission
To determine if plants have boxwood dieback, send plants showing early stages of disease development to a diagnostic clinic. If practical, send the whole plant, including roots.
Branches and twigs that have been dead for several months do not generate accurate diagnosis. Collect three to four 1-foot long symptomatic twigs with both healthy and diseased tissue from recently infected areas.
Wrap twigs individually in dry paper towels and then pack them in plastic ziplock bags. Include a completed sample submission with the sample and send it your state plant diagnostic laboratory. Consult your state plant diagnostic lab before collecting and shipping samples.
Credits
Author: Raj Singh, Associate Professor, Plant Diagnostic Center, LSU AgCenter, Baton Rouge, LA
References
Crouch, J. (2012). Colletotrichum fungal pathogens and symbionts of ornamental nursery and landscape plants. Phytopathology, 102, 104.
Farr, D. F., & Rossman, A. Y. (2016). Fungal Databases, Systematic Mycology and Microbiology Laboratory, ARS, USDA. Retrieved August 12, 2016, from https://nt.ars-grin.gov/fungaldatabases/
Hawk, T., Rhodes, S. C., McBride, S., & Ong, K. (2017). First report of boxwood dieback caused by in Texas. Plant Disease, 102, 242. https://doi.org/10.1094/PDIS-06-17-0821-PDN
Holcomb, G. E. (1967). Pathogenicity of a Colletotrichum sp. isolated from boxwood. Phytopathology, 57, 461.
Singh, R., Graney, L., & Williamson, M. (2015). First report of boxwood dieback caused by Colletotrichum theobromicolain the United States. Plant Disease, 99, 1274. https://doi.org/10.1094/PDIS-09-14-0948-PDN.
USDA National Agricultural Statistics Service. (2019). Quick Stats. Govt. Press, Washington DC.
Weir, B. S., Johnston, P. R., & Damm, U. (2012). The Colletotrichum gloeosporioides species complex. Studies in Mycology, 73 (1), 115-180. https://doi.org/10.3114/sim0011
This work is supported by the Crop Protection and Pest Management Program, project award no. 2022-70006-38001, from the U.S. Department of Agriculture’s National Institute of Food and Agriculture.
Any opinions, findings, conclusions, or recommendations expressed in this publication are those of the speakers and should not be construed to represent any official USDA or U.S. government determination or policy.
For information about the Pest Alert program, please contact the North Central IPM Center at northcentral@ncipmc.org.
Content updated: September 2022
