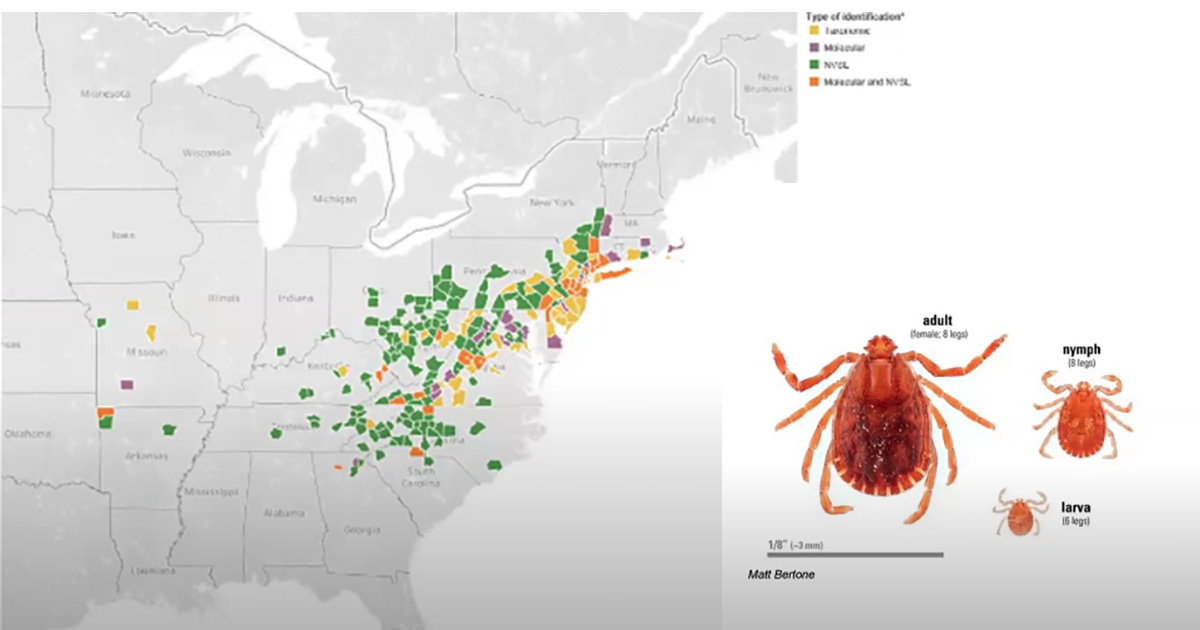
The North Central IPM Center shares news about IPM tools, resources, new technology and trends. Connect with us by
- Subscribing to the Central Issue newsletter
- Joining monthly “Pests and Progress” webinars
- Listening to the “I See Dead Plants” podcast
- Following us on Twitter
- Connecting on LinkedIn